Description
ID# 2221
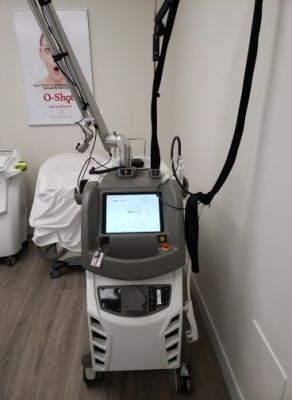
Quanta Systems
MDK 1 EVO
Q-Switch Nd:YAG (long & short pulsed Nd:YAG)
Mfg. 1/20017
Includes: 2x2, 3x3, 4x4mm Handpieces, 2-5mm Adjustable Handpiece, 10mm / 12mm / 14mm Handpieces, Contact Cooling Handpiece, Skin Cryo Handpiece, Footpedal, Key, Eyewear and Manuals.
Multi-Application Dual Wavelength - Two Lasers One System
The Quanta MDK 1 EVP is made up of 2 lasers:
Q-Switched Nd:YAG - Articulated Arm Delivery (Included)
Type: High Peak Power Q-Switched Laser Fractional Spot Sizes: 2x2, 3x3, 4x4Wavelengths: 532nm, 1064nm
Nd:YAG - Fiber Delivery (Included)
Type: Long Pulse & Short Pulse Nd:YAGSpot Sizes
1064: 14mm, 12mm & 10mm & VariableSpot 2-5mm Spot Sizes
532: Variable 532/1064 Variable Handpiece Spot Sizes: 2mm, 3mm, 4mm, 5mm
Wavelength: 532nm, 1064nm Sapphire Contact Cool Handpiece
System is performing to manufacture specifications.
Owners are closing Spa Business.
Mfg. Notes:
The EVO Platform is intended for use in aesthetics, cosmetic and surgical applications requiring incisions, excision, ablation, vaporization and coagulation of body soft tissues in the medical specialties of dermatology, general, plastic and oral surgery as follows:
Indications for use:
1064 & 532nm (Q-Switch, nanosecond mode)
The EVO Platform is intended for treatment of vascular lesions, pigmented lesions, and for hair, tattoo removal and the incision, excision, ablation, vaporization of soft tissues for General dermatology such as, but not limited to treatment of:
532nm (Q-Switched, nanosecond mode)
Removal of light ink (red, sky blue, green, tan, purple and orange) tattoos
Treatment of vascular lesions including, but not limited to:
- Port wine birthmarks
- Telangiectasias
- Spider angiomas
- Cherry Angiomas
- Spider nevi
Treatment of benign pigmented lesions including, but not limited to:
- café-au-lait birthmarks
- Ephalides, solar lentigines
- senile lentigines
- Becker’s nevi
- freckles
- common nevi
- nevus spilus
- Ota Nevus
Treatment of seborrheic keratosis
Treatment of post inflammatory hyperpigmentation
Skin resurfacing procedures for the treatment of acne scars and wrinkles.
1064nm (Q-Switched, nanosecond mode)
Removal of dark ink (black, blue and brown) tattoos
Removal of benign pigmented lesions including:
- nevus of Ota
- Café-au-lait spot
- Ephalides, solar lentigo (lentigines)
- Becker Nevus
- Nevus spilus
Treatment of common nevi
Removal or lightening of unwanted hair
Skin resurfacing procedures for the treatment of acne scars and wrinkles
1064 nm (non Q-Switched- free running mode)
Removal of unwanted hair, for stable long term or permanent hair reduction and for treatment of PFB. The laser is indicated for all skin types, Fitzpatrick I-VI, including tanned skin.
Photocoagulation and hemostasis of pigmented and vascular lesions, such as, but not limited to port wine stains, hemaongiomae, warts, telangiectasiae, rosacea, venus lake, leg veins and spider veins.
Coagulation and hemostasis of soft tissue.
Treatment of wrinkles.
Treatment of mild to moderate inflammatory acne vulgaris.
532 nm (picosecond mode)
Indicated for the removal of tattoos for Fitzpatrick skin types I-III to treat the following tattoo colors: red, yellow and orange.
Indicated for benign pigmented lesions removal for Fitzpatrick skin types I-IV.
1064 nm (picosecond mode)
Indicated for the removal of tattoos for all skin types (Fitzpatrick skin types I-VI) to treat the following tattoo colors: black, brown, green, blue and purple.
Indicated for benign pigmented lesions removal for Fitzpatrick skin types I-IV.
1064 nm (pulsed)
Dermatology/Plastic Surgery:
Intended for the coagulation and hemostasis of benign vascular lesions such as, but not limited to, port wine stains, hemangiomas, warts, telangectasia, rosacea, venus lake, leg veins, spider veins and poikiloderma of civatte; and treatment of benign cutaneous lesions such as warts, scars, striae and psoriasis.
The laser is also intended for the treatment of benign pigmented lesions such as, but not limited to, lentigos (age spots), solar lentigos (sun spots), cafe au lait macules, seborrheic keratoses, nevi, chloasma, verrucae, skin tags, keratosis and plaques.
The laser is also indicated for the treatment of wrinkles such as, but not limited to, periocular and perioral wrinkles.
The laser is also indicated for the treatment of facial wrinkles.
Additionally, the laser is indicated for the treatment of pseudofolliculitis barbae (PFB) and for stable long-term, or permanent hair reduction. Permanent hair reduction is defined as long-term stable reduction in the number of hairs re-growing when measured at 6, 9 and 12 months after the completion of a treatment regime.
It is indicated for the reduction of red pigmentation in hypertrophic and keloid scars where vascularity is an integral part of the scar.
The laser is also indicated for pigmented lesions to reduce lesion size, for patients with lesions that would potentially benefit from aggressive treatment, and for patients with lesions that have not responded to other laser treatments.
It is indicated for use on all skin types (Fitzpatrick I-VI ) including tanned skin, and the removal and permanent reduction of unwanted hair in Fitzpatrick I-VI, including suntanned skin types.
532 nm (pulsed)
Intended for the coagulation and hemostasis of vascular lesions.
Dermatology/Plastic Surgery:
For photocoagulation and hemostasis of vascular and cutaneous lesions in dermatology including but not limited to the following general categories: vascular lesions [angiomas, hemangiomas (port wine), telangiectasia (facial or ex-tremities telangiectasias, venous anomalies, leg veins]; benign pigmented lesions (nevi, lentigines, chloasma, cafe au- lait, tattoos (red and green ink), verrucae, skin tags, keratoses, plaques, cutaneous lesion treatment (hemostasis, color lightening, blanching, flattening, reduction of les10n size).
FDA requires all listings in this category to include the following text: "The sale of this item may be subject to regulation by the U.S. Food and Drug Administration and state and local regulatory agencies. If so, do not bid on this item unless you are an authorized purchaser. If the item is subject to FDA regulation, we will verify your status as an authorized purchaser of this item before shipping of the item."
DISCLAIMER: Regardless of the origin of the equipment, documentation provided or identification appearing upon the equipment, the equipment described and offered here is in no way certified for, recommended for, or offered for any specific use. The purchaser agrees that the seller shall not be held responsible or liable for any injuries or damages, whether incidental or consequential, associated in any way with the equipment. The purchaser, by bidding on this equipment, indicates their acknowledgment of, and agreement to the terms of this disclaimer.